The water Denver Water delivers to customers is lead-free. And for decades, Denver Water has protected its customers from the effects of customer-owned lead-containing water service lines and household plumbing in a variety of ways, including adjusting the pH of the water it delivers to customers.
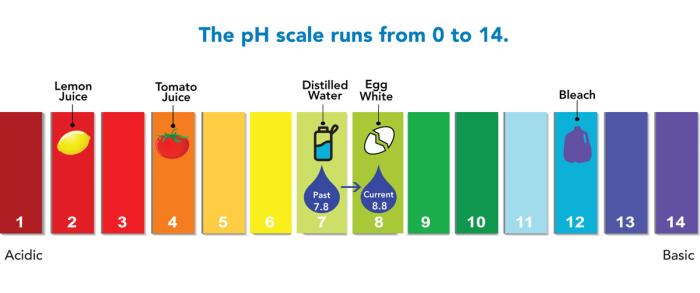
In March 2020, Denver Water increased the pH level of the water it delivers to customers as part of its Lead Reduction Program. The pH level of drinking water reflects how acidic or basic it is; it is measured on a scale of 0 to 14, with 7 considered neutral, meaning there’s a balance between the water’s acidity and basicity.
As part of the Lead Reduction Program, Denver Water slightly increased the pH of the water it delivers, targeting a pH of 8.8. This change was made to reduce the corrosivity of the water, which helps protect customers who have plumbing in their homes that contains lead, such as customer-owned water service lines that connect their home to the Denver Water’s main delivery pipe in the street, solder that connects sections of pipe in their home, and faucet parts.
What you need to know
- Increasing the pH of water is a proven technique used by many water utilities to protect customers from the likelihood of lead or other metals getting into drinking water.
- For years, the water Denver Water delivered to customers had a pH that ranged between 7.5 to 8.5, with a target of 7.8. Since mid-2020, as part of the Lead Reduction Program, Denver Water's pH range is between 8.5 and 9.2, with a target of 8.8.
- Raising the pH of drinking water makes the water less corrosive. This change also strengthens an existing protective coating on the interior of the pipe. The coating reduces the likelihood of lead getting into the water as it passes through customer-owned water service lines, household plumbing and faucets that contain lead.
- Denver Water’s pH management is performing well with lead levels continuing to decline since the pH change was implemented. Lead sampling at properties with known lead service lines has maintained an average below 4.0 ppb.
- Officials with Colorado Department of Public Health and Environment say there are no health concerns about drinking water with pH levels at 8.8.
- Denver Water has worked with irrigation and landscape experts locally and nationwide to learn more about potential impacts from the pH increase. Their feedback is that in general there should not be any major impacts on plants and landscapes. Since this change was implemented in 2020, Denver Water hasn’t observed any noticeable impacts to trees/plants/grass as a result of the pH change.